SCR vs SNCR: Space Velocity, Temp Window & NH3 Slip Explained for Optimal Emission Control

Selective Catalytic Reduction (SCR) and Selective Non-Catalytic Reduction (SNCR) both target nitrogen oxides (NOx) emissions, but they take different paths—especially when it comes to space velocity, temperature window, and ammonia slip.
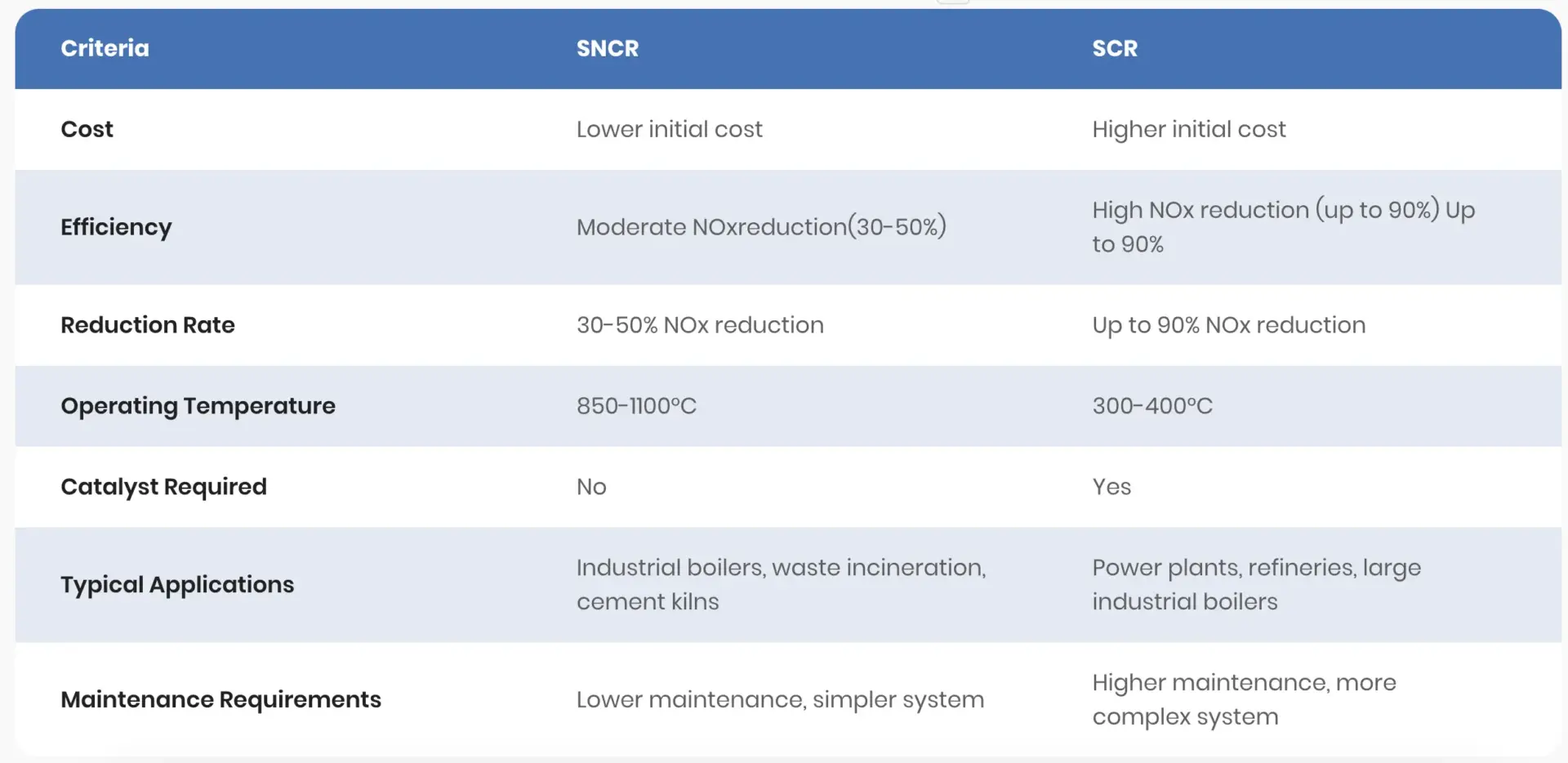
SCR works with a catalyst, converting NOx and ammonia into harmless nitrogen and water. This process usually hits efficiency rates above 95%. SNCR, on the other hand, injects reagents right into the flue gas, skipping the catalyst. That makes SNCR less efficient and a lot more sensitive to temperature swings.
Space velocity and temperature matter a lot. They shape NOx removal rates and how much unreacted ammonia—ammonia slip—escapes into the environment.
Space velocity in SCR controls how much time flue gas spends with the catalyst. That directly impacts NOx conversion and ammonia slip. SNCR’s sweet spot is a narrow temperature window, usually 900°C to 1100°C. Step outside that, and performance tanks—either ammonia decomposes at high temps or reactions crawl at low ones.
Ammonia slip isn’t just a technical detail. Excess ammonia can cause headaches for the environment and mess with overall efficiency.
Fundamental Differences Between SCR And SNCR

SCR and SNCR both aim for lower NOx emissions, but their methods couldn’t be more different. These differences show up in how they work, their efficiency, and where they fit in industry.
SCR uses a metal-based catalyst to boost the reaction between a reducing agent—usually ammonia or urea—and NOx in the flue gas. The catalyst lets NOx turn into nitrogen and water efficiently, usually at 250°C to 450°C.
SNCR skips the catalyst, injecting ammonia or urea right into the hot flue gas above 800°C. The reaction happens on its own, but only within a tight temperature window. Miss the mark, and unwanted ammonia emissions (NH3 slip) go up fast.
The presence or absence of a catalyst shapes everything—temperature ranges, reaction rates, and how much ammonia each process needs.
SCR usually achieves 70-95% NOx removal. The catalyst speeds up the reaction and works at lower temperatures, making it easier to control emissions and minimize ammonia slip.
SNCR typically cuts NOx by 30-50%. Performance depends on keeping temperatures between 850°C and 1100°C. Drop too low and NOx conversion suffers; go too high and ammonia slip or side reactions spike.
Comparison Table:
| Attribute | SCR | SNCR |
| NOx Reduction Efficiency | 70-95% | 30-50% |
| Operating Temperature | 250°C – 450°C | 850°C – 1100°C |
| Catalyst Use | Yes | No |
| Ammonia Slip | Low | Higher, harder to control |
SCR tends to win out in places with strict emission limits, thanks to higher NOx reduction and better ammonia slip control.
SCR gets the nod when strict NOx reduction and ammonia slip limits are on the table. Large plants—think power stations, refineries, and cement factories—use SCR where flue gas temperatures stay steady around 300-400°C.
But that performance comes at a price. SCR systems need more capital and maintenance due to catalysts and complex gear.
SNCR keeps things simple and cheaper. It works for very hot flue gases and less strict emission rules. Boilers and furnaces often use SNCR, especially where catalysts aren’t practical or budgets are tight.
Selection comes down to a few factors:
- Flue gas temperature profile
- NOx and ammonia emission limits
- Capital and operating costs
- Space and maintenance needs
Some recent strategies even combine SNCR and SCR to balance cost and performance, hitting ultra-low NOx and keeping ammonia slip manageable.
Impact Of Space Velocity In SCR And SNCR
Space velocity sits at the heart of SCR and SNCR efficiency. It shapes NOx reduction, sets residence time for flue gas, and can make or break ammonia slip levels.
Space velocity refers to the volume of flue gas moving through the catalyst or reaction zone per unit catalyst volume per hour. Usually measured in hr⁻¹, SCR folks call it Gas Hourly Space Velocity (GHSV).
High space velocity means flue gas rushes through, with less time to react. Too high, and NOx conversion drops. Too low, and reactors get big and costly. The trick is balance—enough time for reactions, but not so slow that the system becomes unwieldy.
SNCR also feels the effect of space velocity, but not as directly. Catalyst volume doesn’t control things here. Instead, temperature and reagent injection timing matter more, along with how quickly flue gas moves.
NOx conversion hangs on space velocity. SCR systems often operate around 2,400 hr⁻¹, giving about 1.67 seconds of residence time. That’s enough for the catalyst to cut NOx by up to 98%, at least in hybrid SNCR/SCR setups.
Push space velocity higher, and residence time shrinks. The catalyst can’t keep up, and NOx conversion falls off. At 6,000 hr⁻¹ and above, efficiency really suffers.
SNCR gets by with higher space velocities, since temperature and mixing drive its reactions. Still, the interplay between space velocity and ammonia slip can’t be ignored. More speed means less time for ammonia to react, and more of it escapes unreacted.
Flue gas makeup, temperature, and flow rate all influence how space velocity plays out. SCR catalysts work best at 300–400°C, and space velocity must match so NOx gets enough time to react with ammonia.
SNCR’s optimal window is higher—850–950°C. Fast flue gas flow demands precise ammonia injection. Variations in flow speed or volume can throw off space velocity, so tuning the system is a must.
Dust, humidity, and contaminants also affect catalyst surface area and reaction rates. Operators need to watch these factors closely to keep space velocity and system performance on track.
Key Entities: NOx reduction, catalyst surface area, residence time, ammonia slip, flue gas temperature.
Temperature Window Optimization

Temperature control makes or breaks NOx reduction in SCR and SNCR. The right range lets ammonia or urea react with NOx efficiently. Miss the window, and incomplete reduction or wasted reagent becomes a problem.
SNCR works best between 850°C and 1100°C. Inside that range, urea or ammonia reacts with NOx to cut emissions.
Drop below 850°C, and reactions slow. NOx removal tanks. Go above 1100°C, and ammonia breaks down, losing effectiveness and sometimes even creating more NOx.
Keeping temperature inside that window matters. Ammonia slip rises fast outside the optimal range, leading to environmental headaches and extra treatment costs. That’s why system designs lean heavily on precise temperature monitoring and control.
SCR uses a different temperature range: usually 300°C to 450°C. Catalysts like vanadium-based or Cu-SSZ-13 do their best work here, converting NOx into nitrogen and water.
Temperatures below 300°C slow down catalyst activity. Above 450°C, catalysts start to degrade, shortening their lifespan and forcing replacements or regeneration.
The catalyst’s temperature window ties directly to residence time and space velocity. Ammonia injection happens downstream of combustion to keep things in the sweet spot and minimize ammonia slip.
Drifting outside these temperature ranges spells trouble. SNCR at over 1100°C sees ammonia decompose, sometimes making more NO instead of less. Under 850°C, reactions crawl, NOx stays high, and ammonia slip increases.
SCR reactors get sluggish at low temperatures and risk catalyst poisoning or sintering at high ones. More ammonia slips through when reactions can’t finish, often requiring extra cleanup downstream.
Uniform temperature in the flue gas and careful control of injection points help keep conditions right for both reagent reaction and catalyst performance.
Understanding And Managing NH3 Slip
Ammonia slip happens when unreacted ammonia escapes from SCR or SNCR systems after trying to cut NOx emissions. Keeping ammonia slip under control matters for system efficiency and meeting tough emission standards.
Injection rates, temperature management, and catalyst performance all shape ammonia slip levels and their downstream impact.
Sources And Causes Of Ammonia Slip
Excess ammonia gets injected to ensure full NOx reduction, but sometimes it doesn’t react and escapes in the exhaust. Uneven ammonia distribution in Selective Catalytic Reduction (SCR) or off-ratio NH3 to NOx conditions usually drive this issue.
Temperature swings play a big role. If operation drifts outside the optimum window, reaction rates drop or ammonia oxidizes, bumping up slip. Gas flow rate—space velocity—affects residence time and how completely NOx turns into nitrogen.
Selective Non-Catalytic Reduction (SNCR) skips the catalyst, so ammonia reacts less efficiently and slip runs higher. Common causes include:
- Overdosing ammonia for NOx reduction
- Low exhaust temperatures slowing reactions
- Poor NH3/flue gas mixing
- Aged or fouled catalyst reducing conversion
Precise ammonia injection and good catalyst health help limit slip.
Impacts On Emissions And System Performance
Ammonia slip doesn’t just waste reagent—it releases unreacted NH3 into the air. This can turn into fine particulates or even re-form nitrogen oxides, undermining emission reduction efforts.
High slip signals incomplete NOx conversion and can corrode equipment, foul plant surfaces, or trigger odor complaints. Dialing in injection rates, keeping the system in the catalytic temperature window (usually 250–450°C), and watching catalyst condition all matter.
- NH3 injection rates
- Operating temperature in the right window
- System flow and catalyst condition
Emission rules often cap NH3 slip at just a few ppm, so precise dosing and monitoring are non-negotiable. Some plants add advanced ammonia slip catalysts (ASC) downstream to trim slip further without hurting NOx control (GlobalSpec, 2024).
Comparative Analysis: NOx Reduction Performance
Selective Catalytic Reduction (SCR) usually delivers higher NOx reduction than Selective Non-Catalytic Reduction (SNCR). Results depend on operating temperature, catalyst shape, and reagent injection method. Each technology’s effectiveness shifts with process conditions, which also affect ammonia slip and reagent use.
Efficiency Across Varying Operating Conditions
SCR works best between 300°C and 450°C, often cutting NOx by over 90%. That temperature window lets the catalyst convert NOx and NH3 into nitrogen and water efficiently.
Go outside that range, and catalyst activity falls off. SNCR needs higher temperatures, typically 850°C to 1100°C, and delivers 30–70% NOx reduction depending on the process. SNCR is more temperature-sensitive and struggles if the gas misses the sweet spot.
Residence time and reagent distribution also matter for SNCR. SCR’s catalyst speeds up reactions, so SCR works at lower temperatures than SNCR, but demands tight temperature control for best results.
One 2023 Valmet report, “NOx reduction with SCR, SNCR and Scrubber methods,” found SCR with good gas flow and temperature management can hold NOx removal above 90%.
Influence Of Catalyst Aging (SCR)
Catalyst aging drags down SCR’s NOx reduction over time. Ash, sulfur, or chemical poisons clog the surface, cutting the number of active sites for reaction.
As catalyst efficiency drops, ammonia slip can creep up because less NOx gets converted—unreacted ammonia slips through. Regular catalyst activity checks keep performance up and prevent excess ammonia emissions.
Periodic testing and evaluation help estimate catalyst life and schedule maintenance. A 2024 Babcock & Wilcox study notes catalyst aging can cut SCR efficiency by 10–15% after 3–5 years, depending on fuel and operating conditions.
Reagent Type And Injection Methods
Ammonia (NH3) and urea show up as the main reagents in both SCR and SNCR. SCR usually runs ammonia for faster, more complete NOx reduction over a catalyst. Urea gets used too but first converts to ammonia before reacting.
Injection method shapes reagent distribution and NOx reduction. SCR injects upstream of the catalyst with tight control, ensuring good mixing and less ammonia slip. SNCR injects directly into hot flue gas zones, skipping catalysts and relying on temperature and residence time.
SCR uses less reagent overall thanks to higher efficiency; SNCR often needs more to get the same NOx cut. Choosing reagent and injection spot means weighing NOx removal, operating cost, and ammonia slip risk.
A 2022 review in “Advancements in SCR technologies for NOx reduction” found hydrocarbon reductants and hydrogen can work in SCR, but cost and handling issues keep ammonia as the go-to reagent.
Hybrid And Integrated SCR/SNCR Systems
Hybrid SCR/SNCR setups blend selective catalytic reduction (SCR) and selective non-catalytic reduction (SNCR) for better NOx reduction, while managing ammonia slip and system limits. SNCR tackles NOx at high temperatures; SCR follows up at a catalytic stage, balancing efficiency, cost, and space.
Designs focus on optimizing process conditions and handling trade-offs like catalyst size and ammonia slip.
Process Design Principles
Hybrid SCR/SNCR systems start with SNCR, injecting urea or ammonia into flue gas at 850–1100°C. This step knocks out much of the NOx but usually leaves some ammonia slip.
After SNCR, flue gas moves through the SCR catalyst (300–400°C). The catalyst knocks down remaining NOx and soaks up leftover ammonia from SNCR, cutting overall slip.
The main design move: run SNCR near its best temperature window for NOx removal, then let SCR clean up slip ammonia and any leftover NOx. This setup means thinner catalyst layers, so capital cost and space drop.
Hybrid systems need careful gas velocity (space velocity) and temperature profile management to keep NOx removal steady and ammonia slip low across both stages.
Advantages And Trade-Offs
Hybrid systems pull off higher NOx reduction efficiency than SNCR alone, sometimes hitting ultra-low emissions for strict rules. They use less catalyst than stand-alone SCR, so initial costs and footprint shrink.
But, hybrid setups bring more complexity—two-stage operation demands precise ammonia injection and tight temperature control to avoid slip or catalyst poisoning. Ammonia slip management stays critical; too much slip causes environmental headaches, while too little cuts NOx control.
Maintenance costs can rise with dual systems, though the ability to retrofit SNCR into SCR plants, or vice versa, adds flexibility.
Summary table of advantages and trade-offs:
| Advantages | Trade-Offs |
| Improved NOx removal efficiency | Increased system complexity |
| Reduced SCR catalyst volume required | Requires precise temperature control |
| Lower capital costs than standalone SCR | Ammonia slip must be carefully managed |
| Flexibility for retrofit projects | Higher maintenance needs |
A 2023 Yara analysis, “Hybrid SNCR/SCR Systems,” calls this approach a cost-performance balance for many industrial NOx control jobs, especially where stricter emission limits apply.
Frequently Asked Questions
SCR and SNCR differ mainly in operating temperature, space velocity, and ammonia slip. These differences shape NOx reduction and system efficiency. Catalyst use, temperature control, and ammonia injection rate all matter.
What is the optimal temperature range for Selective Catalytic Reduction (SCR) systems?
SCR systems run best from 300°C to 400°C. This window lets the catalyst convert NOx to nitrogen and water efficiently. Temperatures outside this range hurt efficiency and can damage the catalyst.
How do the space velocities compare between SCR and Selective Non-Catalytic Reduction (SNCR) processes?
SCR handles higher space velocities thanks to the catalyst’s faster reaction rates. SNCR works at lower space velocities, relying on direct chemical reactions. This shapes reactor size and gas flow management.
What are the typical ammonia slip rates for SCR and SNCR technologies?
SCR can keep ammonia slip very low—often under 2 mg per normal cubic meter. SNCR usually sees higher slip, since controlling ammonia without a catalyst is trickier and can lead to more unreacted ammonia in emissions.
Can you explain the importance of the catalyst in SCR reactions?
The catalyst in SCR lowers the energy barrier, speeding up NOx and ammonia reactions. It enables NOx reduction at lower temperatures than SNCR. Catalyst quality and regular maintenance shape overall system performance and durability.
What factors influence the efficiency of NOx reduction in SNCR applications?
SNCR efficiency leans hard on the right temperature—usually 900°C to 1100°C. Too much heat breaks down ammonia, hurting NOx reduction. Uneven reagent distribution and poor temperature control also drag down effectiveness.
How does ammonia injection rate affect the performance of SCR and SNCR systems?
SCR systems demand tight control of ammonia injection. The goal is to cut NOx emissions without causing too much ammonia slip.
SNCR technology also depends on precise ammonia dosing. The process works best when the dose matches the ideal temperature window, maximizing the reaction and keeping excess ammonia out of the exhaust.
Poor control in either setup leads to real efficiency losses. Both SCR and SNCR systems need careful management to hit their targets.
